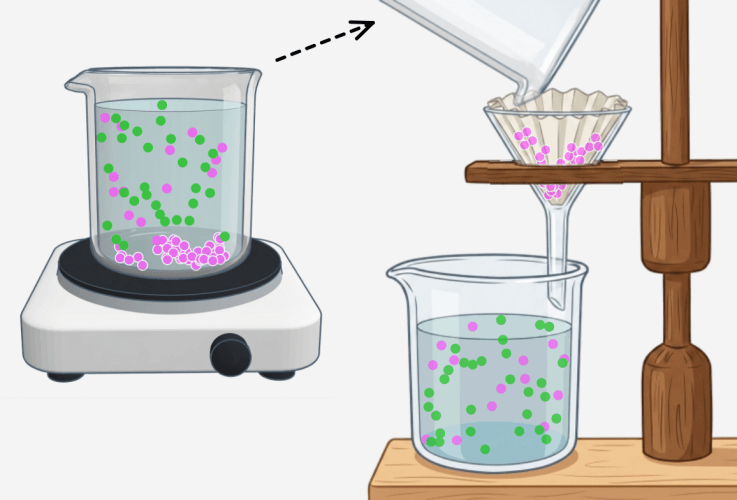
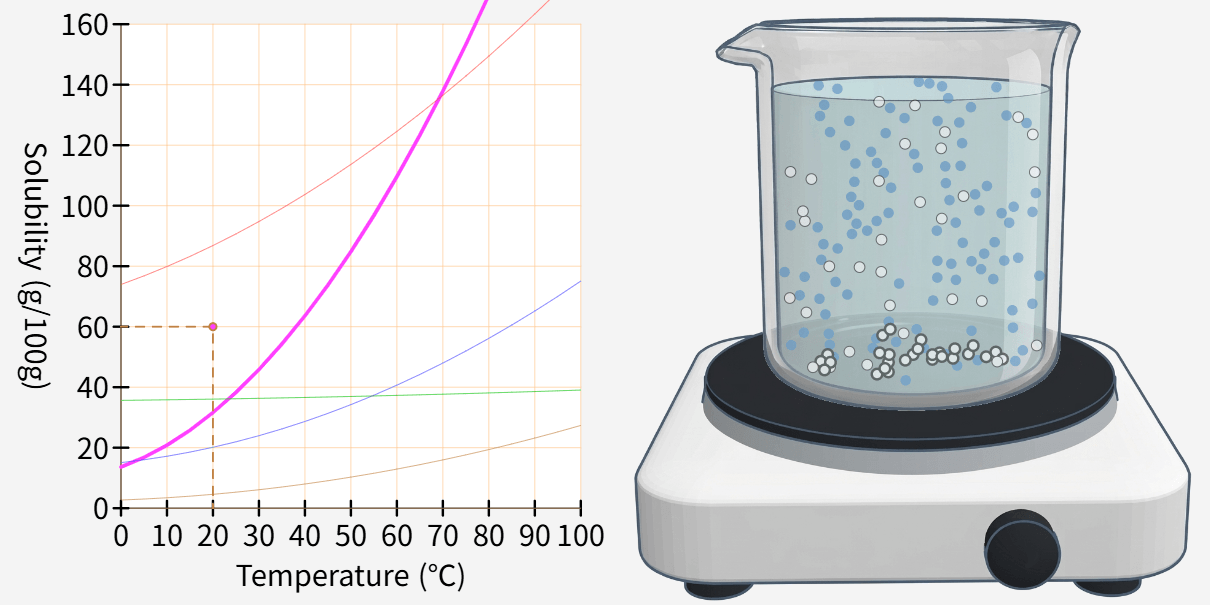
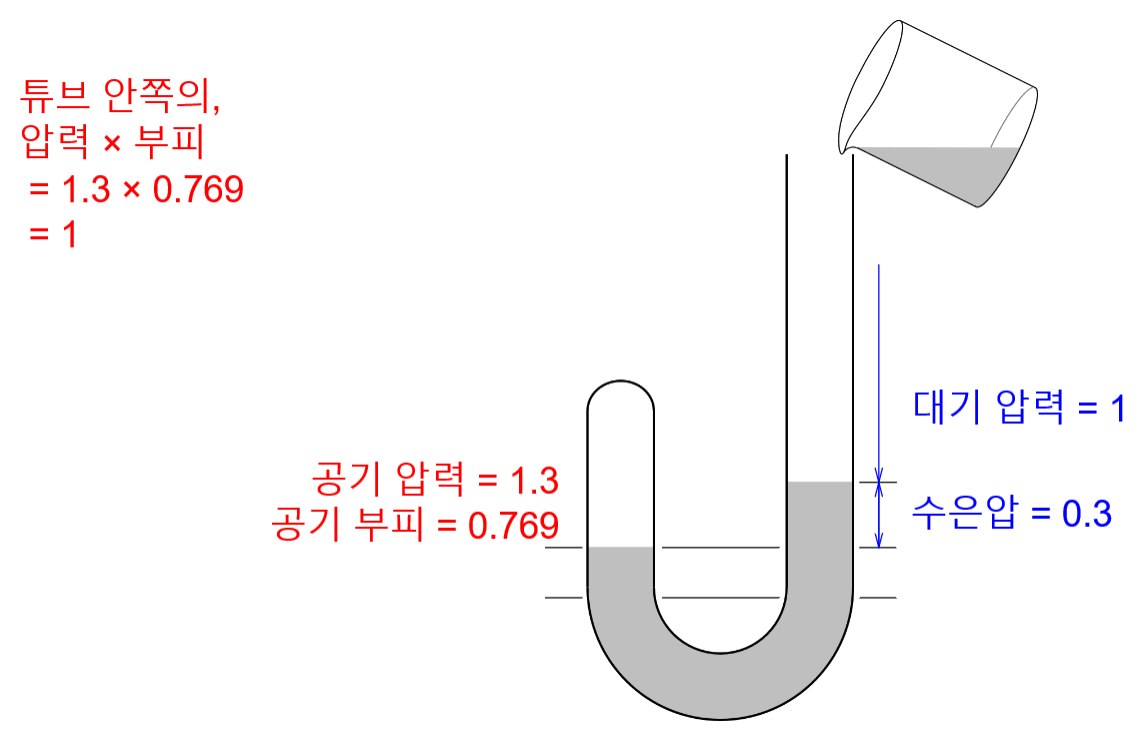
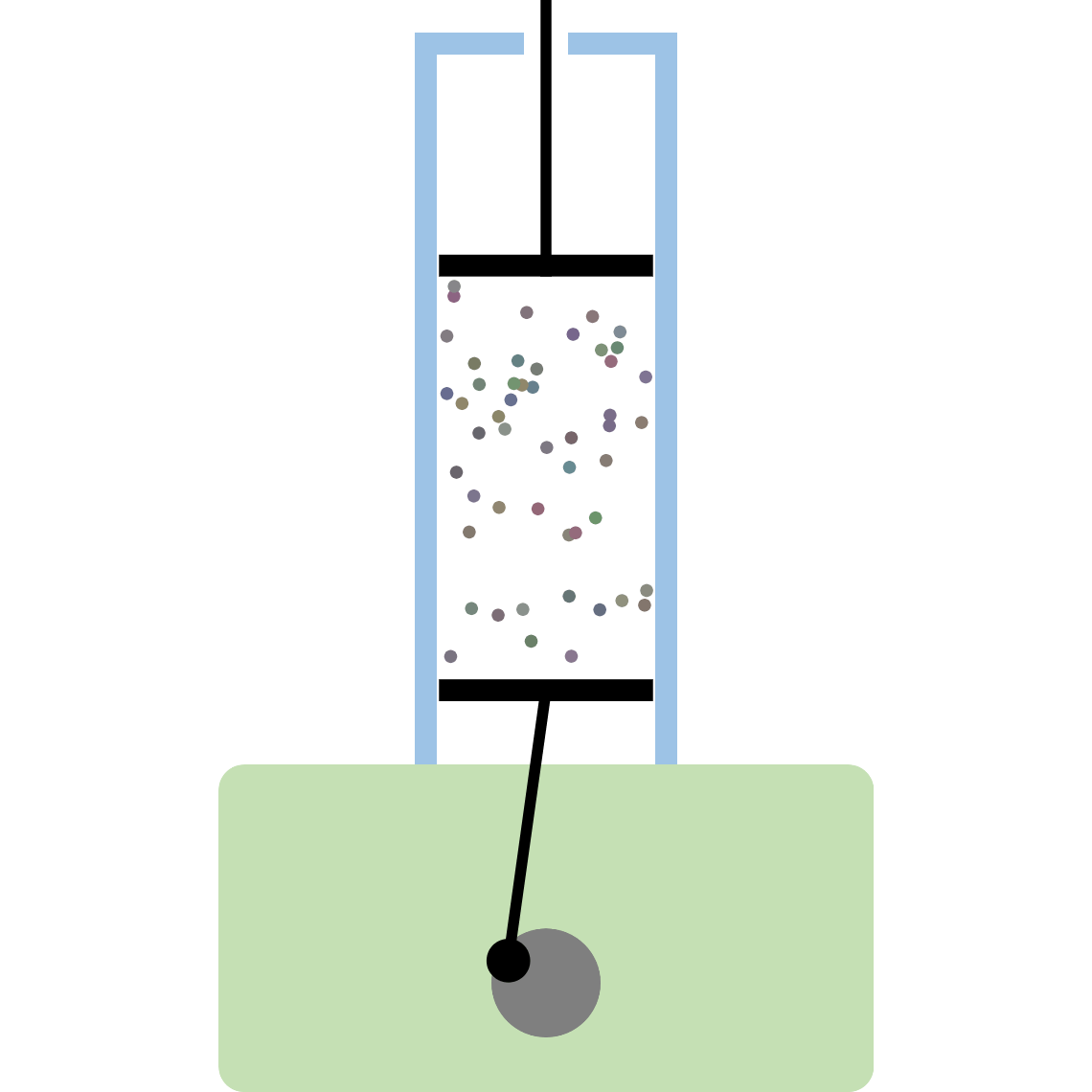
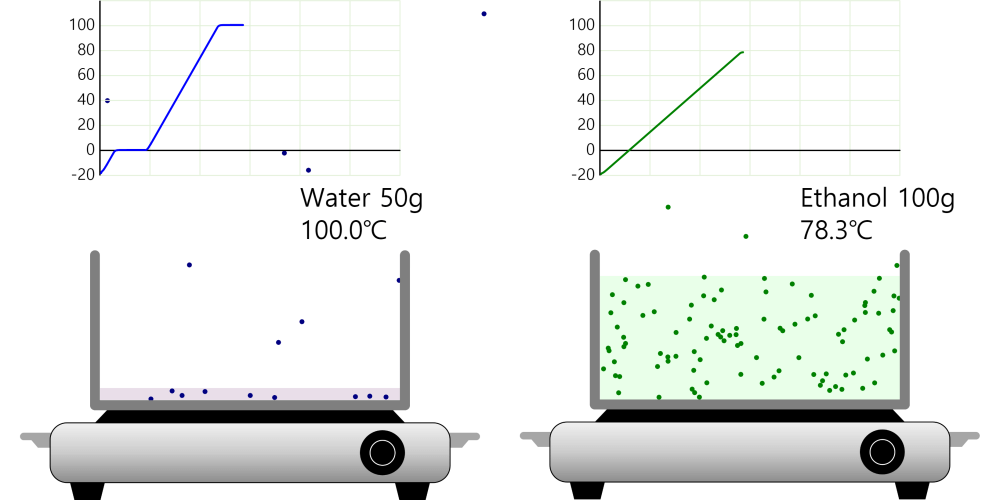
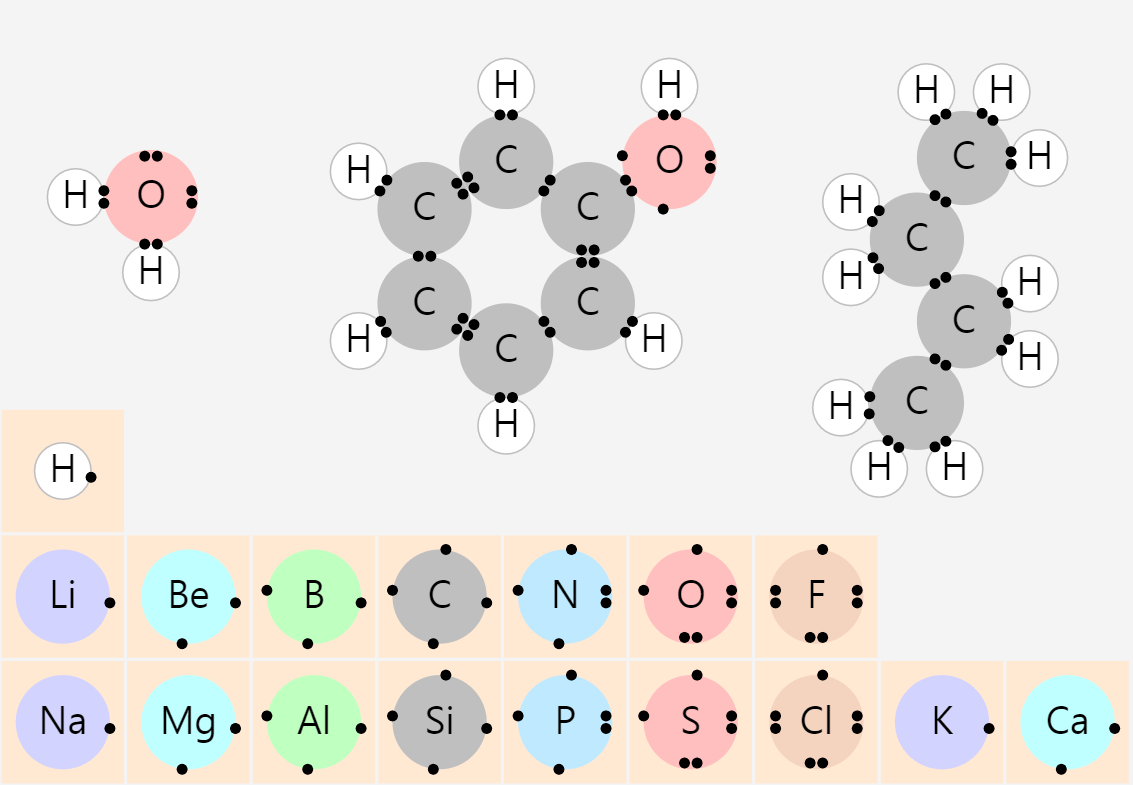
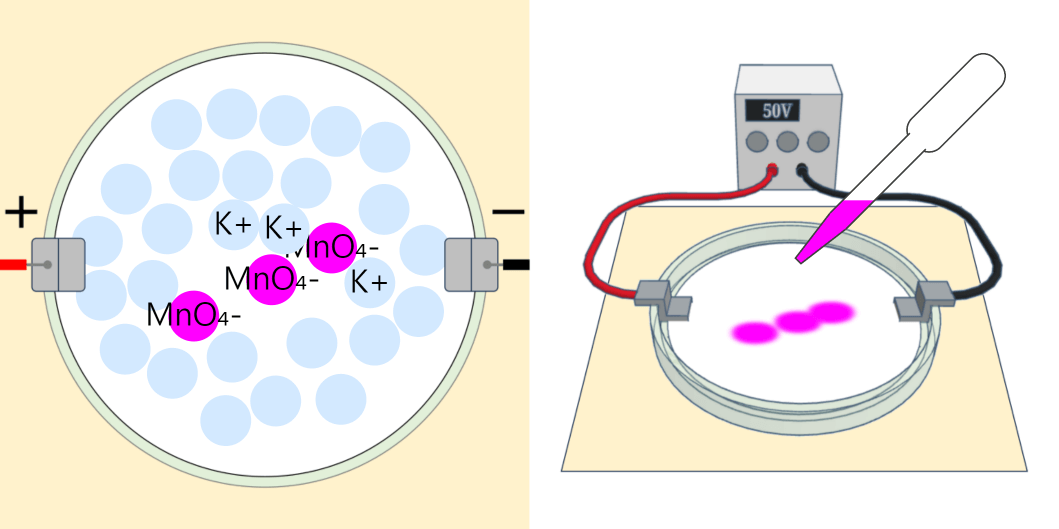
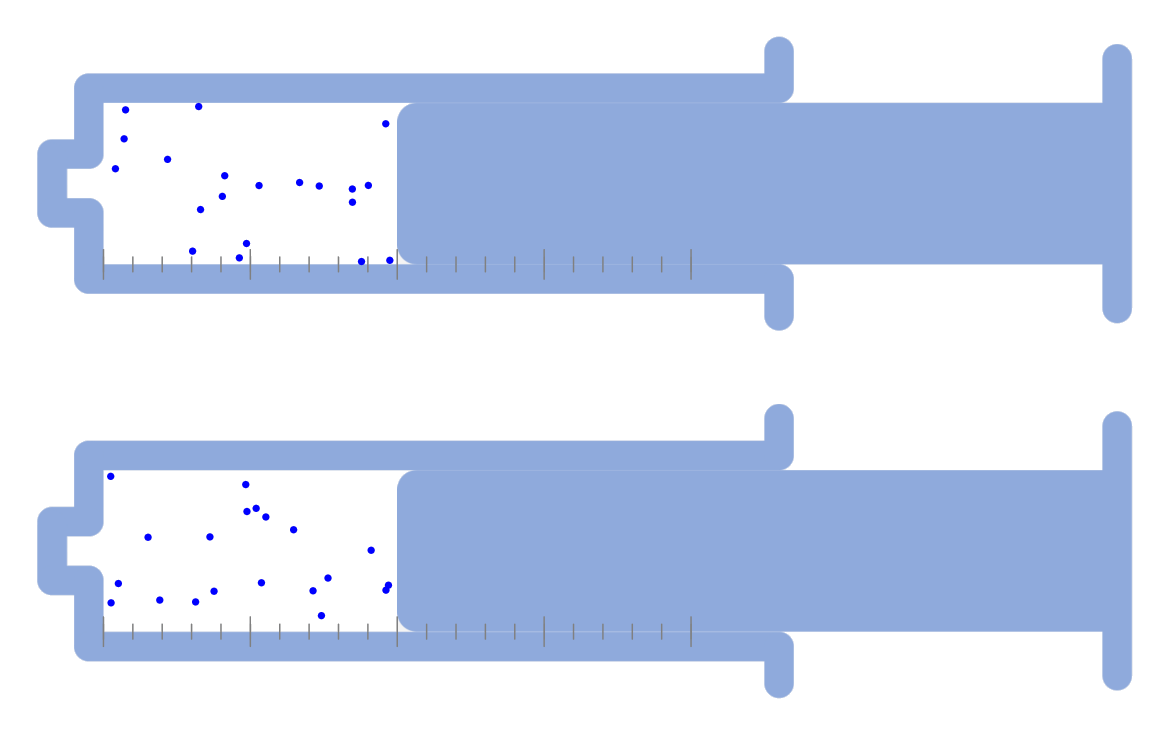
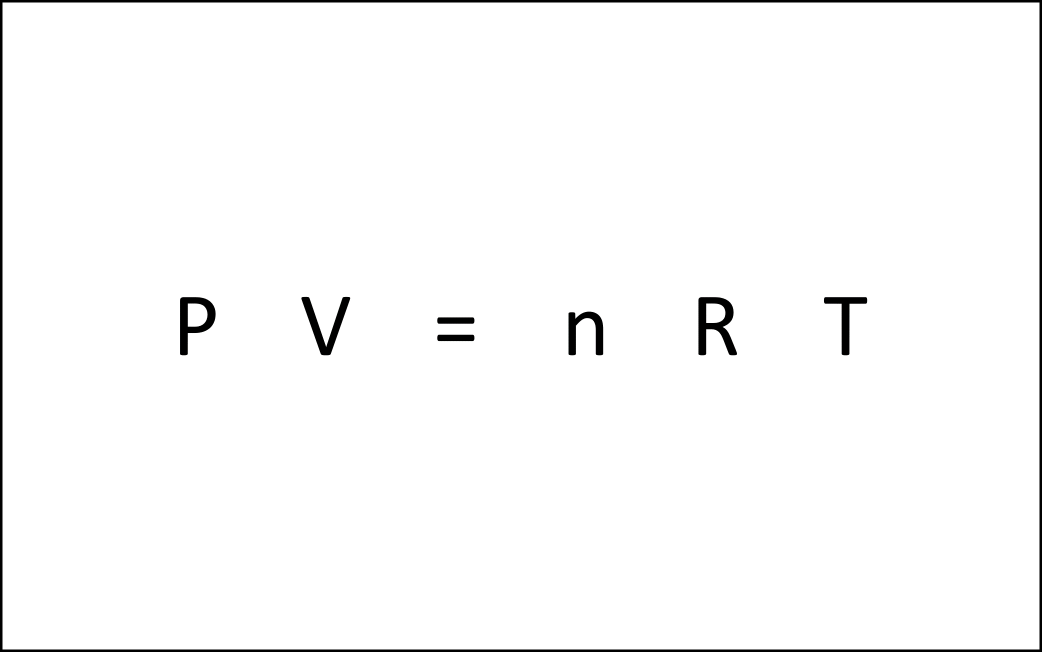Recrystallization (Principle of precipitation)
Lower the temperature to precipitate some of the substances as a solid, then filter them through filter paper. Recrystallization Recrystallization is a method of separating and purifying a substance by using the property that the amount … more