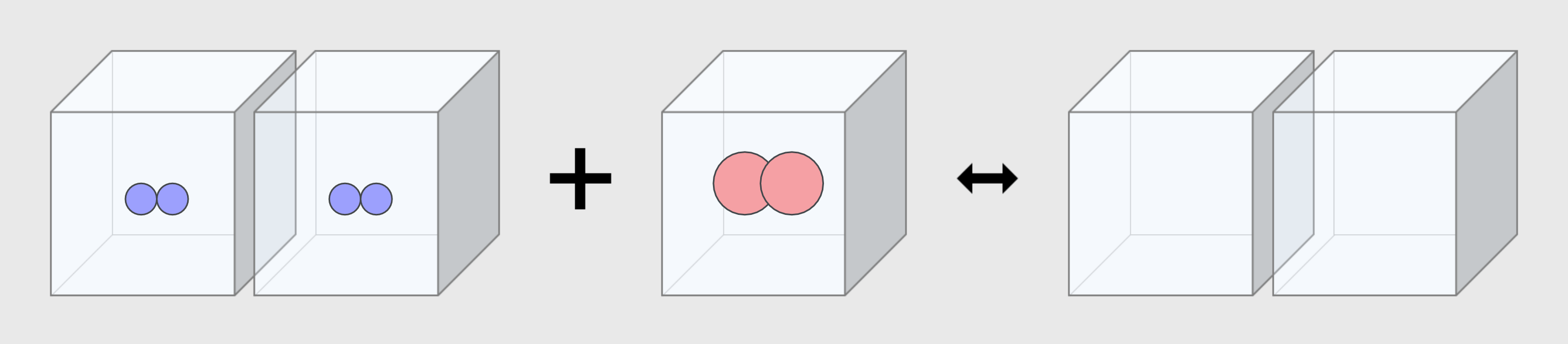
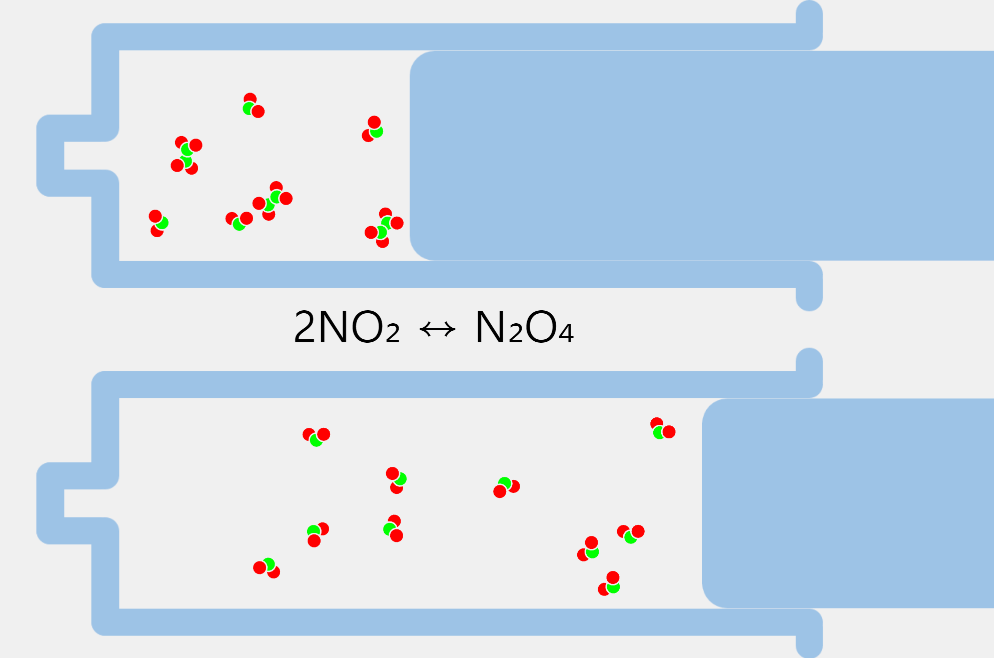
State of Water 2
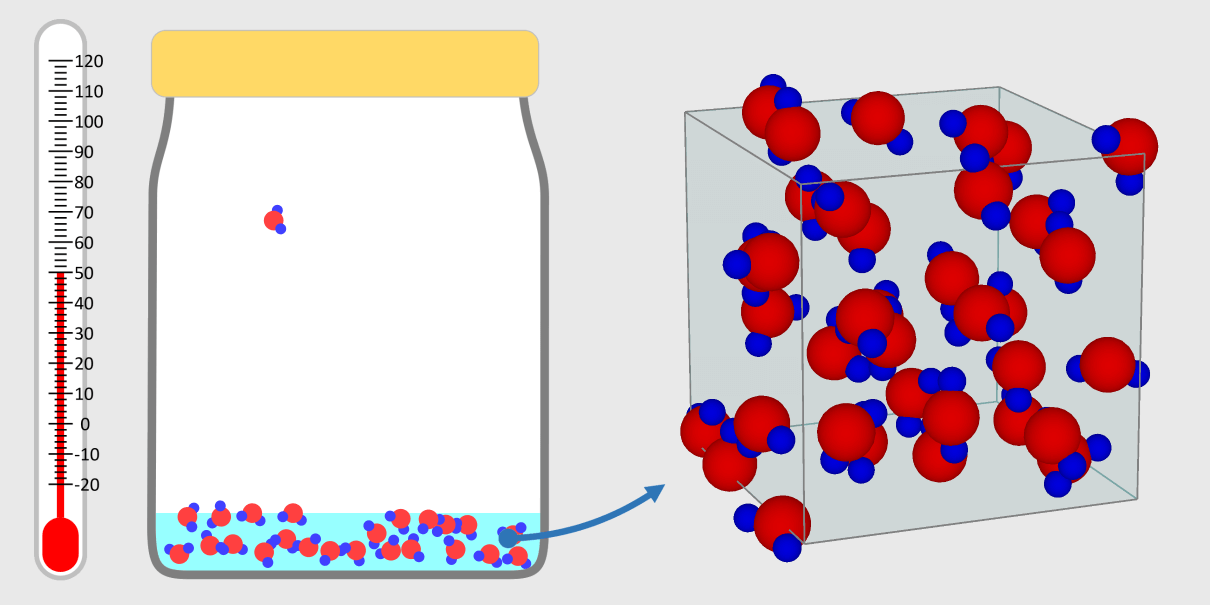
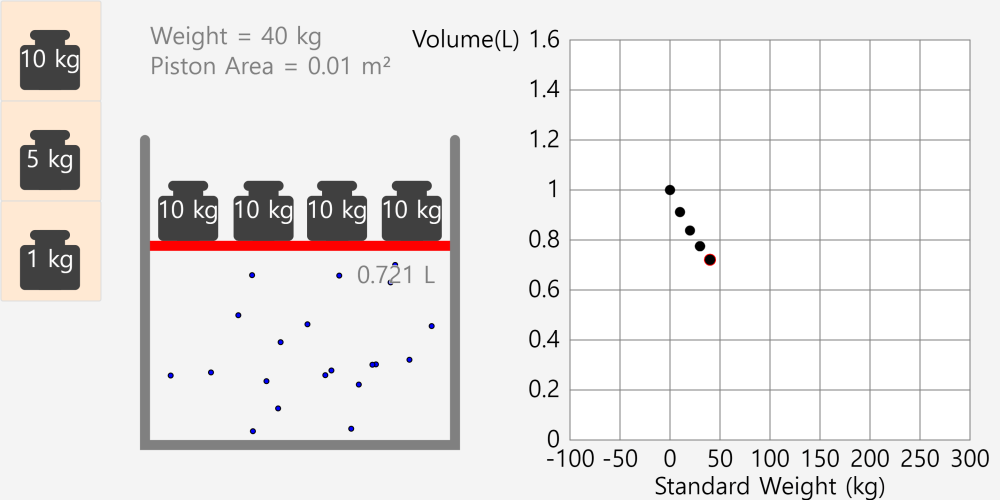
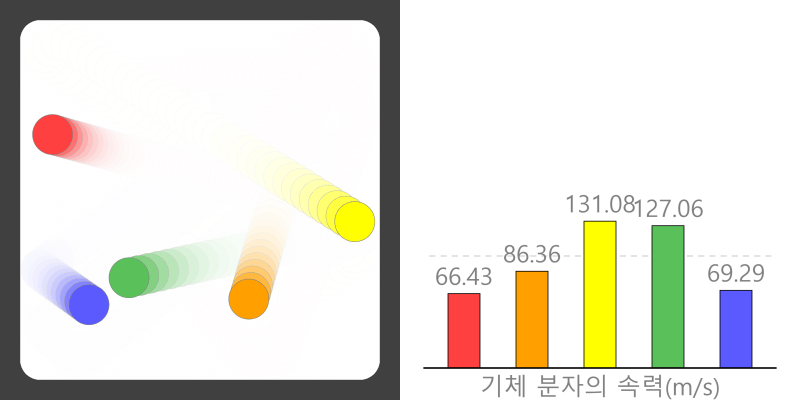
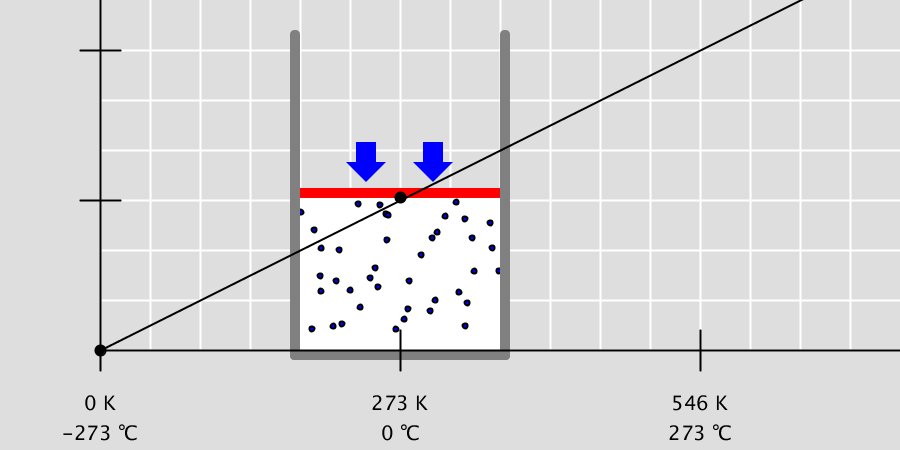
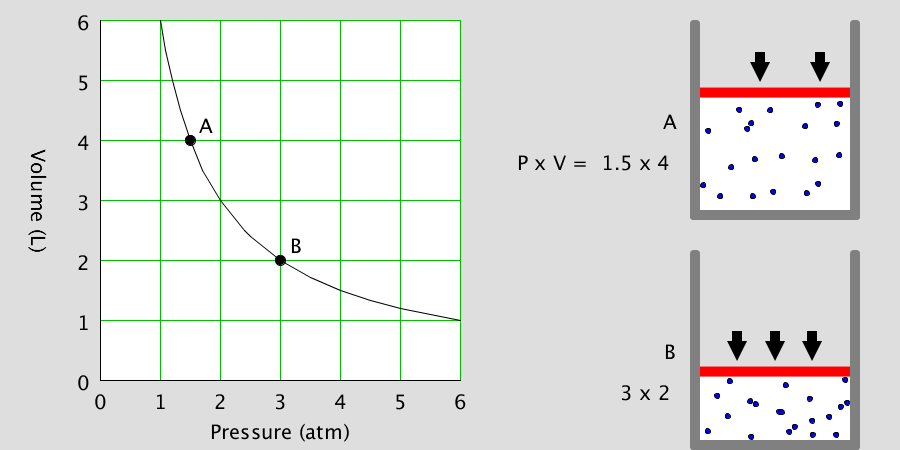
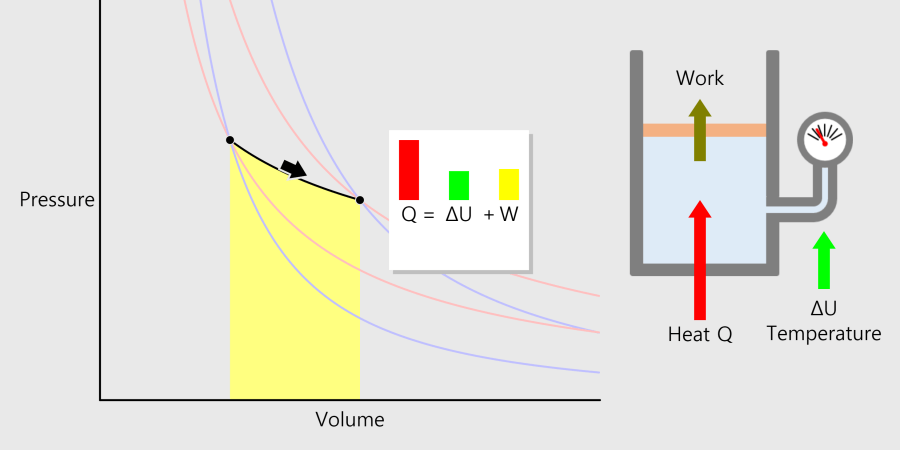
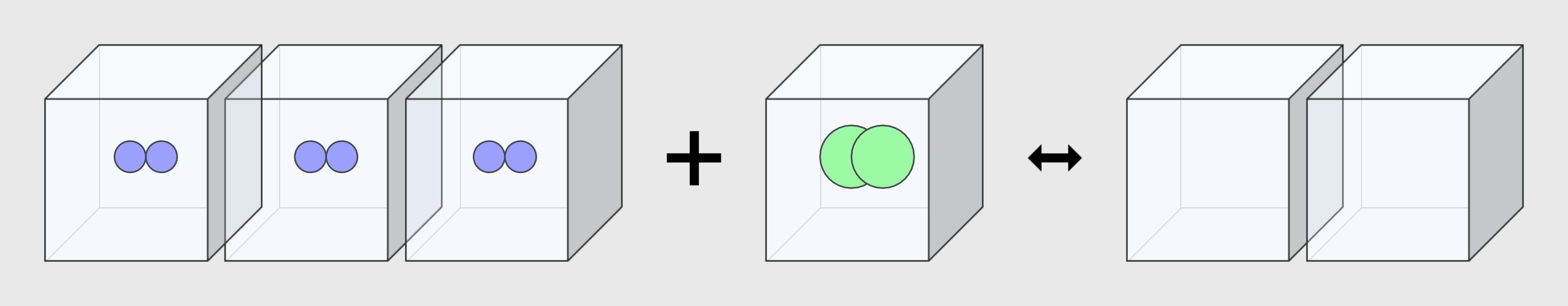
* By adjusting the temperature of the thermometer, you can observe the state of the water corresponding to the temperature. * Depending on the temperature of the water, some water molecules may evaporate. State of matter The material around us … more