Light has both particle and waves properties at the same time. Likewise, de Broglie (France, 1892 – 1987) thought that such things as electrons that had so far been considered particles might possess wave nature (1932). This is the idea of the de Broglie waves, or de Broglie’s matter waves.
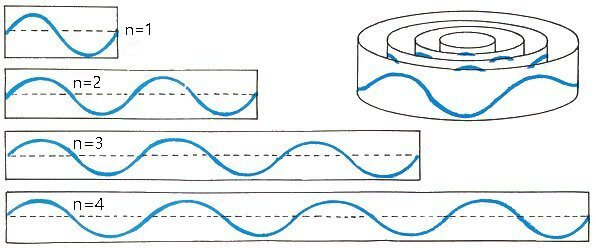
Let us consider a hydrogen atom with Bohr’s quantum theory. In that theory, the quantum condition to determine the atom’s stationary states seems to claim that the circumference of the orbit of the electron revolving about the nucleus should be an integral multiple of the wavelength of the de Broglie wave of the electron.
This quantum condition says that the de Broglie wave of the electron must be a continuous standing wave around the nucleus.

The de Broglie wave does not connect smoothly in the right figure because the circumference is not an integral multiple of the wavelength. So, the right figure does not a correct de Broglie wave.